January 23, 2026
By Caleb Carter, E.I.T.; Daniel J. Nord, P.E.
www.utieng.com
What is Distillation?
Distillation is a physical process used to purify a substance by evaporating one substance from a mixture. This vapor is then condensed and collected by cooling the vapor back into a liquid, called the distillate. The distillate is not 100% pure following a distillation process, as it typically contains various impurities present in the initial solution as well as some of the solvent. Generally, the distillate can be further distilled to increase purity (Clifton, 2024). Figure 1 below shows a typical lab scale distillation apparatus set up.
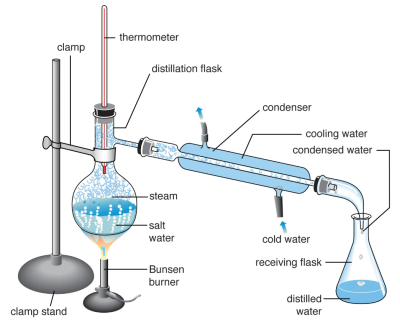
Figure 1 – Typical distillation apparatus set up for a lab
Note. A laboratory distillation apparatus demonstrating desalination of water. From “Distillation summary,” by The Editors of Encyclopaedia Britannica, n.d., Encyclopædia Britannica (https://www.britannica.com/summary/distillation). Copyright 2026 by Encyclopædia Britannica, Inc.
Separations Theory 101
In order to distill a substance from a mixture, it is important to understand the physical properties of each. The pure component of the distillate has a higher vapor pressure than the other components in the mixture. This means that the component with the highest vapor pressure has the lowest boiling point and is most likely to evaporate as the temperature of the mixture increases (Clark, 2023). To determine the boiling point ranges at various mixture compositions, a T-xy diagram is used. The T-xy diagram shows the saturation curves for the liquid and vapor phases across varying temperature and mixture compositions. This is used to determine the liquid phase and vapor phase compositions at any given temperature or vise versa. Figure 2 below shows a typical T-xy diagram for benzene.
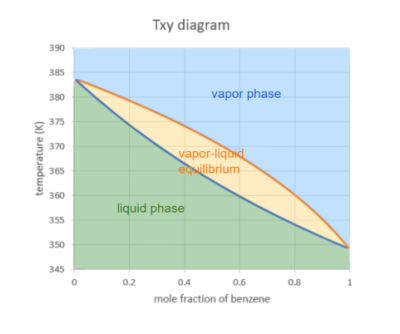
Figure 2 – T-xy diagram for benzene at 1 atm
Note. From “Txy Diagram,” in Foundations of Chemical and Biological Engineering I, by J. Verrett, 2020, BCcampus (https://pressbooks.bccampus.ca/chbe220/chapter/txy-diagram/). CC BY-SA 4.0.
Distillation Columns
A distillation column is an industrial scale up from the lab apparatus. Towards the top of the column there is a condenser used to change the vapor back to the liquid phase (called the tops), where it can be sent downstream for further processing. Towards the bottom of the column is a reboiler, used to evaporate the liquid that pools at the bottom of the column. A portion of this is recovered in the liquid phase (called the bottoms) and sent downstream for further processing. Each of the tops and bottoms streams are much purer in their respective substances than the feed. Figure 3 below shows a typical distillation column schematic.
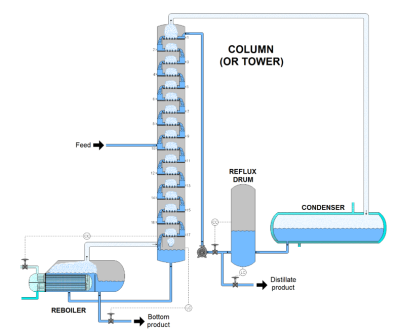
Figure 3 – Typical distillation column schematic
Note. Schematic diagram of a continuous distillation column. From “The ultimate guide to distillation and distillation columns,” by Sophie, 2024, Gusta Water (https://www.gustawater.com/blog/distillation-column.html). Copyright 2024 by Gusta Water.
Design Considerations
Design of a distillation column is a very tedious process to do by hand. Software simulations are typically utilized now to assist with design. Common software packages include ASPEN Plus and ChemCAD. These are powerful software packages used to provide process results, such as internal mass and energy balances, physical properties, height of the column, width of column, number of theoretical trays, etc. even when dealing with non-ideal and multicomponent mixtures. Figure 4 below show what a typical distillation column set up would look like in ASPEN Plus. The physical properties of substances can be found on a few different databases, such as DECHEMA or Dortmund Database, which are often included in simulation software packages. This is used with the specified thermodynamic model to size columns. Binary or multicomponent VLE data can be regressed in simulation software to produce the thermodynamic parameters required.
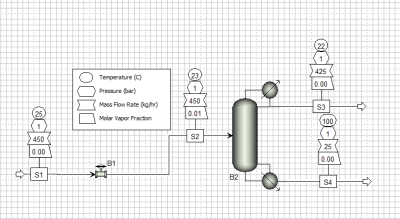
Figure 4 – Typical distillation column set up in ASPEN Plus
Column internals include an option of either trays or packing, and which one is used is dependent on the process. There are many different designs for trays, but they all are a similar function. There are perforations of varying design that allow the vapor to flow upwards to the next stage, while the liquid flows across the tray and eventually downward to the next tray (Porter, 2011). Figure 5 below shows an example of a sieve tray, commonly used in trayed distillation columns.

Figure 5 – A single pass sieve tray
Note. Photograph of a single pass sieve tray (DN 1600) in stainless steel. From “Sieve trays,” by Kempro, n.d., Kempro (http://kempro.eu/sievetrays.html). Copyright by Kempro.
Packing is another option when it comes to designing distillation column internals. Packing is a solid material that is used to enhance the interaction between the liquid and vapor phase. There are two different kinds of packing that can be used in a distillation column – structured and random. Structured packing is a geometrically arranged sheet or grid that is installed inside the column. It offers lower pressure drop, high capacity, and improved mass transfer efficiency than random packing. It is also generally more expensive than random packing. Random packing is irregularly shaped materials randomly dumped in the column. It provides a large surface area for mass transfer and is generally cheap (Marvi, 2023). Figure 6 shows some general examples of the different types of packing.

Figure 6 – Structured and random packing examples
Note. A comparison of structured and random packing used in natural gas dehydration towers. From “Gas absorber tower: How the 3 types work to dehydrate your natural gas,” by K. Andrews, n.d., Kimray (https://kimray.com/training/gas-absorber-tower-how-3-types-work-dehydrate-your-natural-gas). Copyright 2025 by Kimray, Inc.
There are several different methods for determining the number of stages in a distillation column. One other specific method for estimating the number of trays in a column is called the McCabe-Thiele shortcut method. This method takes the specific VLE data from the process and combined with the desired purities in the outputs determines the number of theoretical stages. It should be noted that this is a simplified method and the assumptions used for this method do not typically apply to real world applications, therefore this method should only be used as a reference point when running simulations or designing a column (Neutrium, 2017). Figure 7 below shows an example of a completed McCabe-Thiele diagram for a column with 6 stages.
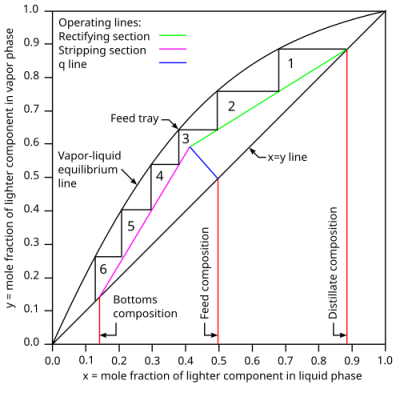
Figure 7 – McCabe-Thiele diagram showing theoretical stages in distillation
Figure 7: McCabe-Thiele diagram showing theoretical stages in distillation. From “McCabe-Thiele diagram,” by Slashme, 2008, Wikimedia Commons (https://commons.wikimedia.org/wiki/File:McCabe-Thiele_diagram.svg). CC BY-SA 3.0.
Reflux is another variable that can be manipulated to change the design of the column. Reflux is the portion of condensed overhead liquid product that is returned to the column. The reflux ratio is the ratio of liquid returning to the column vs leaving the column. Reflux improves the ability of the column to separate, allowing multiple iterations of vaporization/condensation, increasing the purity of the distillate. The minimum required reflux ratio, Rmin, is a mathematically calculated variable that yields an infinite number of stages. The optimum reflux ratio in distillation is the balance point where the total cost (capital + operating) is minimized, and is typically 1.1 to 1.3 times the minimum reflux ratio. The reflux ratio has a direct impact on the number of stages, height, width, and overall costs of the column. A higher ratio reduces the number of trays, but increases column diameter, condenser and reboiler size, heating/cooling loads, while a lower ratio does the opposite, creating a trade-off that defines the optimum point for efficient, economical separation (Porter, 2011).
Other parameters include the feed location, operating pressure of the column, and the types of reboilers and condensers. The feed location for the column should be the point in the column where the composition of the liquid/vapor in the column closely matches the composition of the feed (Moshfeghian, 2018). The operating pressure of the column depends on the specific vapor liquid equilibrium data of the mixture, and the P-xy diagram should be consulted. Finally, expected duty for the reboilers and condensers can be calculated from the simulation software, and used as a basis of design for the reboilers and condensers for the column.
Common Problems
There occasionally arises some issues during operation of distillation columns. Some typical problems that might be seen is flooding, foaming, entrainment, and weeping/dumping. Flooding is when trays overfill with liquid while foaming is when the trays overfill with vapor. Entrainment is when liquid is carried by vapor up to the tray above, while weeping/dumping is when the liquid begins to leak through the perforations in the trays. These are all common problems that greatly affect the ability of the column to separate. Issues that arise can usually be detected through process monitoring. These issues specifically will often cause changes in the pressure drop across trays and can be detected with proper monitoring. Low vapor flow can be caused with issues on the reboiler, while liquid flow is directly related to the reflux ratio, i.e. the amount of reflux returning to the column (Tham & Costello, 2023).
Typical Control Schemes
It is important to monitor the distillation column for product quality purposes and for longevity of the equipment. Some typical controls include temperature, composition, pressure, level, and flow controls. These are all used together to strategically monitor every aspect of the column and the process to ensure everything is operating as designed.
References
Clark, J. (2023, January 30). Fractional distillation of non-ideal mixtures (azeotropes). Chemistry LibreTexts. https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Equilibria/Physical_Equilibria/Fractional_Distillation_of_Non-ideal_Mixtures_(Azeotropes)
Clifton, J. (2025, May 9). What is the distillation process?: The chemistry blog. ReAgent Chemical Services. https://www.chemicals.co.uk/blog/what-is-the-distillation-process
Marvi, M. (2023, May 26). The difference between random packing & structured packing. Mach Engineering. https://www.machengineering.com/blog/random-packing-vs-structured-packing/
Moshfeghian, Dr. M. (2018, February 1). Optimum feed tray location in an NGL fractionation column. PetroSkills. https://www.petroskills.com/en/blog/entry/feb18-fac-optimum-feed-tray-location-in-an-ngl-fractionation-column#:~:text=The%20tray%2Dby%2Dtray%20rigorous,M=14%20including%20reboiler).
Neutrium. (2017, April 17). McCabe-Thiele Plot. Neutrium. https://neutrium.net/articles/unit-operations/distillation/mccabe-thiele-plot/
Porter, E. A. (2011, February 2). Distillation. THERMOPEDIA. https://www.thermopedia.com/content/703/
Tham, M. T., & Costello, R. C. (2023). Factors affecting distillation column operation. Distillation an introduction. https://www.rccostello.com/distil/distilop.htm
Images
Figure 1: The Editors of Encyclopaedia Britannica. (n.d.). A laboratory distillation apparatus demonstrating desalination of water [Diagram]. Encyclopædia Britannica. Retrieved January 22, 2026, from https://www.britannica.com/summary/distillation
Figure 2: Verret, J., Qiao, R., & Barghout, R. A. (2020). Txy diagram. In Foundations of Chemical and Biological Engineering I. BCcampus. https://pressbooks.bccampus.ca/chbe220/chapter/txy-diagram/
Figure 3: Sophie. (2024, September 20). The ultimate guide to distillation and distillation columns. Gusta Water. https://www.gustawater.com/blog/distillation-column.html
Figure 5: Kempro. (n.d.). Sieve trays. Retrieved January 23, 2026, from http://kempro.eu/sievetrays.html
Figure 6: Andrews, K. (n.d.). Gas absorber tower: How the 3 types work to dehydrate your natural gas. Kimray. https://kimray.com/training/gas-absorber-tower-how-3-types-work-dehydrate-your-natural-gas
Figure 7: Slashme. (2008). McCabe-Thiele diagram [Diagram]. Wikimedia Commons. https://commons.wikimedia.org/wiki/File:McCabe-Thiele_diagram.svg